Research Article
Seasonal Variations in Species Composition and Community Structure of Zooplankton in a Two Perennial Lakes of Coimbatore, Tamil Nadu, Southern India
Narasimman Manickam1,2*, Periyakali Saravana Bhavan2 and Perumal Santhanam1
1Marine Planktonology & Aquaculture Laboratory, Department of Marine Science, Bharathidasan University, Tiruchirappalli-620 024, Tamil Nadu, India
2Crustacean Biology Laboratory, Department of Zoology, Bharathiar University, Coimbatore-641 046, Tamil Nadu, India
*Corresponding author: Narasimman Manickam, Marine Planktonology & Aquaculture Laboratory, Department of Marine Science, Bharathidasan University, Tiruchirappalli 620024, Tamil Nadu, India, E-mail: nmanickam5@gmail.com; drmanickam5@gmail.com
Citation: Manickam N, Bhavan PS, Santhanam P (2017) Seasonal Variations in Species Composition and Community Structure of Zooplankton in A Two Perennial Lakes of Coimbatore, Tamil Nadu, Southern India. J Aquat Res Mar Sci 2017: 1-12. doi: https://doi.org/10.29199/ARMS.101013
Received Date: 9 October, 2017; Accepted Date: 23 October, 2017; Published Date: 12 April, 2018
Abstract
The present investigation was carried out in selected lakes of Coimbatore: Sulur and Ukkadam, Tamil Nadu, Southern India. The seasonal variations of zooplankton species composition and diversity were studied for a period of two years from March-2012 - February-2014, on monthly interval basis of four seasons like summer, pre-monsoon, monsoon and post-monsoon. Totally 29 species of zooplankton, which include 11 species of Rotifera, 10 species of Cladocera, 7 species of Copepoda and 6 species of Ostracoda were recorded and identified from Sulur lake and 28 species of zooplankton comprising 10 species of Rotifera, 8 Cladocera, 6 Copepoda and 4 Ostracoda were recorded in Ukkadam lake. The Rotifera was found to be predominant with 34% followed by crustacean zooplankton of Cladocera (31%), Copepoda (25%) and Ostracoda (10%) at Sulur lake while in Ukkadam lake for Rotifera were found to be predominant with 35% followed by Cladocera (30%), Copepoda (27%) and Ostracoda (8%). The population density of zooplankton recorded at Sulur lake was ranged between 51,895 and 1,07,505 ind./L and in Ukkadam lake it was 89,385 and 1,89,435 ind./L. Zooplankton population was found to be higher in summer months (March to May-2013) while lower in monsoon months (September to November-2012) and intermediate for numbers were recorded during post-monsoon season in the Sulur and Ukkadam lakes. The present result revealed that the zooplankton productivity was high. Therefore, it could be continuously utilized for the inland aquaculture purposes if properly water quality management measures are adopted in the lakes ecosystem. Hence at regular intervals monitoring of water quality, proper maintenance and lake management measures should be adopted by public and governmental organizations to sustain these ecosystems for future generations.
Keywords: Biodiversity; Crustacean Zooplankton; Rotifera; Sulur Lake; Ukkadam Lake
Introduction
Freshwater zooplankton plays a main role in ponds, lakes and reservoirs ecosystem with food chain of the aquatic ecosystem [1,2]. Zooplankton communities play an important role in the function of aquatic ecosystems by providing linkages in food webs through consuming primary and small secondary production and providing food to higher trophic consumers [3-6]. The zooplankton species were eating millions of little algae, bacteria and minute invertebrates which may otherwise grow out-of-control state. As filter feeders, a community of zooplankton can filter through the volume of an entire lake in a matter of days. Especially zooplankton is more important live feed source of many omnivorous and carnivorous of fin-fish and shell-fish and support the necessary amount of high nutrients of protein for the larval growth. Zooplankton is sensitive to changes in aquatic environment and has been suggested to be good biological indicators for water quality, lake trophic state, and types of water mass [7-9].
The zooplankton has belonging to four major groups of including Rotifera and crustacean zooplankton of Cladocera, Copepoda and Ostracoda. They are highly sensitive to environmental variation of water quality, as a result change in their abundance, species composition, species diversity and community structure can provide important indication of environmental change or disturbance. Moreover, due to their short life cycle, these communities often respond quickly to environmental change. Zooplankton offer several advantages as indicators of environmental quality in both lakes and rivers: as a group, they have worldwide distribution and the species composition and community structure are sensitive for changes in environmental conditions, nutrient enrichment [10,11] and different levels of pollution [12]. In many lakes all over the world, the eutrophication has caused a drastic change in the biological structure, the disappearance of submerged macrophyte and the regime shift of aquatic ecosystem [13,14]. Eutrophication is a kind of nutrient enrichment process of water body, which often results in an excessive growth of phytoplankton, an extremely low transparency and a significant decrease in species diversity [15,16]. During the past centuries, lake eutrophication, as a worldwide environmental problem, has been a serious threat to aquatic organism survival and drinking water safety of surrounding residents [17], and a major cause of concern in the developing countries [18,19] like China, India, Bangladesh, Pakistan etc., as well as the developed countries [16,20,21] like European countries, USA, Canada etc. Through these researches, vast nutrients into lakes, especially phosphorus and nitrogen, have been proved to be the major cause of lake eutrophication and degradation of aquatic ecosystem [22]. The observation of present study is to investigate seasonal variations, species composition, percentage composition, population density, species diversity, species richness and evenness of zooplankton from the two perennial lakes.
Materials and Methods
Study area
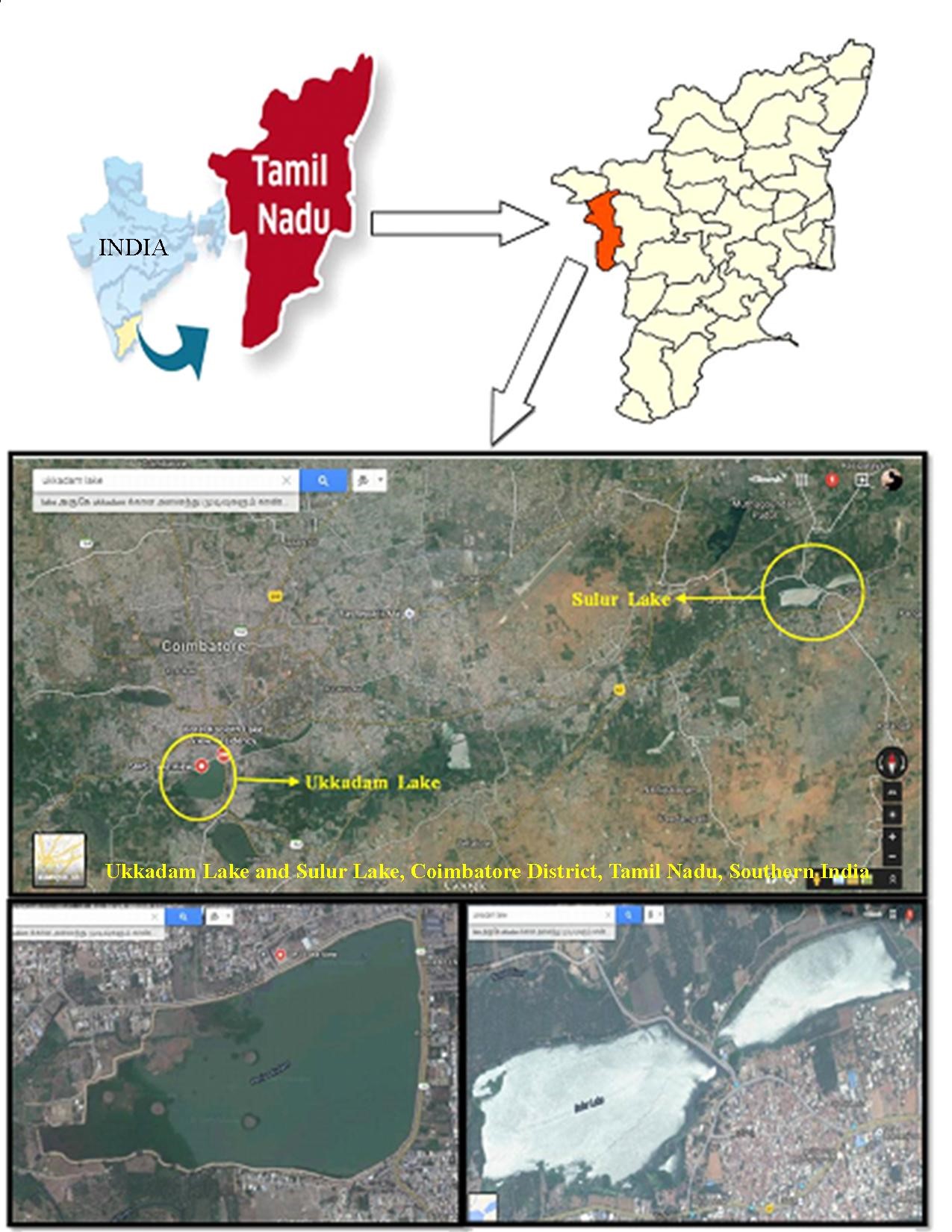
The two freshwater perennial lakes namely Sulur (Station-1; Lat: 11º01' N; Long: 77º07' E) and Ukkadam (Station-2; Lat: 10º59' N; Long: 76º57' E) in Coimbatore District, Southern India were selected for present investigation. Two sampling sites were fixed in each lake as shown in figure 1 and samplings were done from March-2012 - February-2014. These lakes were brought under Department of Fisheries, Govt. of Tamil Nadu, India, and mainly used for culturing common carps such as Catla catla, Labeo rohita and Cirrhinus mrigala. Some aquatic plants like algae and other water grasses are commonly seen in the lakes. A number of migratory birds are encountered in the lakes during winter season.
|
Figure 1: Satellite view of study area. |
Qualitative and quantitative analysis of plankton
The plankton samples were collected during early morning between 5.00 and 6.00 AM, at first week of the month. The zooplankton samples were collected using Towing-Henson’s standard plankton net (150 µm mesh) by towing horizontally at surface for about 10 minutes with uniformly 10 km speed of boat. For the quantitative analysis100 liters of water were filtered through a plankton net made up of bolting silk (No: 10, mesh size: 150 µm) using a 10-liter capacity plastic container. After filtering out the water, the plankton biomass was transferred to specimen bottles containing 5% of neutralized formalin and subjected to microscopic analysis. Zooplankton is segregated group wise were separated under a binocular stereo zoom dissection microscope using a fine needle and brush. Individual species of plankton was mounted on microscopic slides on a drop of 20% glycerin after staining with eosin and rose bengal.
The identification of zooplankton was made referring the standard manuals, text books and monographs [23-27] using a compound microscope and photomicrographs were taken using, Inverted Biological Microscope (Model Number INVERSO 3000 (TC-100) CETI) attached a camera (Model IS 300). The 1 ml of sample was taken with a wide mouthed pipette and poured into the counting cell of the Sedg-wick counting Rafter following Santhanam et al., [28] and counted under light microscope. Species diversity index (H) was calculated using Shannon and Weaner’s formula [29]; H1 =pi ∑ log2 pi, I = 1s, Where, H1 → species diversity in bits of information per individual, pi - ni / N (proportion of the sample belonging to the species), ni → Number of individual in all the sample; Species Richness (SR) was calculated as described by Gleason [30]; D = 1 - C, Where, C = ∑ pi2, pi - ni/N, ni – N/S, N → Total number of individuals, S → Number of species in the collection; Evenness index (J1) was calculated by using the formula of Pielous (1966); J1 = H1/log2 s, Where, H1 = species diversity in bits of information per individual, S = Number of species. Shannon and Weaner’s species diversity index (H'), Species Richness (SR), and Evenness index (J) were analysed using the PAST (Palaeontological Statistics), software (ver. 2.02).
Results
Species composition (Qualitative analysis)
Totally 34 species of zooplankton were recorded in the Sulur lake (Tables 1 and 2) and (Figures 2 and 3). Off these, 11 species were contributed by Rotifera (3 families and 4 genera), 10 Cladocera (4 fand 6 genera). A total of 28 species of zooplankton were recorded from the Ukkadam lake,milies and 6 genera), 7 Copepoda (2 families and 5 genera) and 6 Ostracoda (1 family a which comprising 10 species of Rotifera (2 families and 3 genera), 8 Cladocera (4 families and 6 genera), 6 Copepoda (2 families and 5 genera) and 4 Ostracoda (1 family and 4 genera).
Table 1: Check list of zooplankton recorded in Sulur and Ukkadam lakes during March-2012 - February-2014. |
Table 2: List of zooplankton species presented in Sulur and Ukkadam Lake during March-2012 - February-2014. |
|
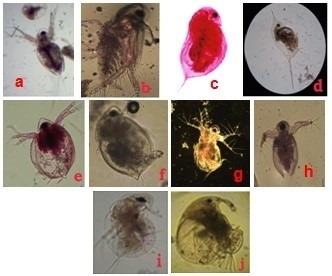
Figure 2: Rotifera recorded from Sulur and Ukkadam lakes. Brachionus angularis (a), Brachionus calyciflorus (b), Brachionus caudatus personatus (c), Brachionus diversicornis (d), Brachionus falcatus (e), Brachionus quadridentatus (f), Brachionus rubens (g), Keratella tropica (h), Asplanchna brightwelli (i), Asplanchna intermedia (j), Filinia longiseta (k) |
|
Figure 3: Cladocera recorded from Sulur and Ukkadam lakes. Diaphanosoma sarsi (a), Diaphanosoma excisum (b), Daphnia carinata (c), Daphnia magna (d), Ceriodaphnia cornuta (e), Ceriodaphnia reticulate (f), Moina brachiata (g), Moina micrura (h), Moinodaphnia macleayi (i), Macrothrix goeldii (j) |
Percentage composition
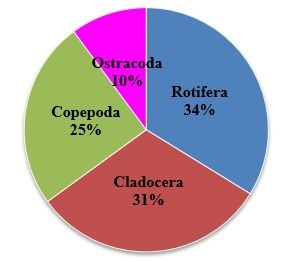
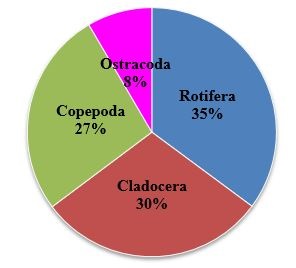
In the present observation, among the zooplankton recorded rotifera holds the top rank in percentage composition at Sulur and Ukkadam lake. The rotifera were found to be predominant with 34% followed by Cladocera (31%), Copepoda (25%) and Ostracoda (10%) at Sulur Lake (Figure 4). At Ukkadam Lake, rotifera were found to be predominant with 35% followed by Cladocera (30%), Copepoda (27%) and Ostracoda 8 % (Figure 5).
|
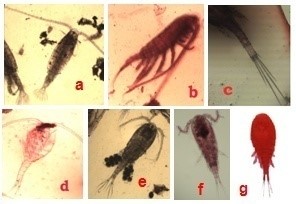
Figure 4: Copepoda recorded from Sulur and Ukkadam lakes. Heliodiaptomus viduus (a), Sinodiaptomus indicus (b), Eucyclops speratus (c), Mesocyclops hyalinus (d), Mesocyclops leuckarti (e), Thermocyclops hyalinus (f), Thermocyclops decipiens (g) |
|
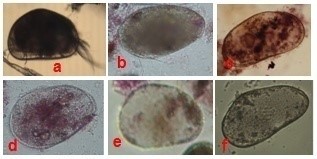
Figure 5: Ostracoda recorded from Sulur and Ukkadam lakes. Cypris protubera (a), Eucypris bispinosa (b), Strandesia elongate (c), Cyprinotus nudus (d), Heterocypris dentatomarginatus (e), Cypretta fontinalis (f) |
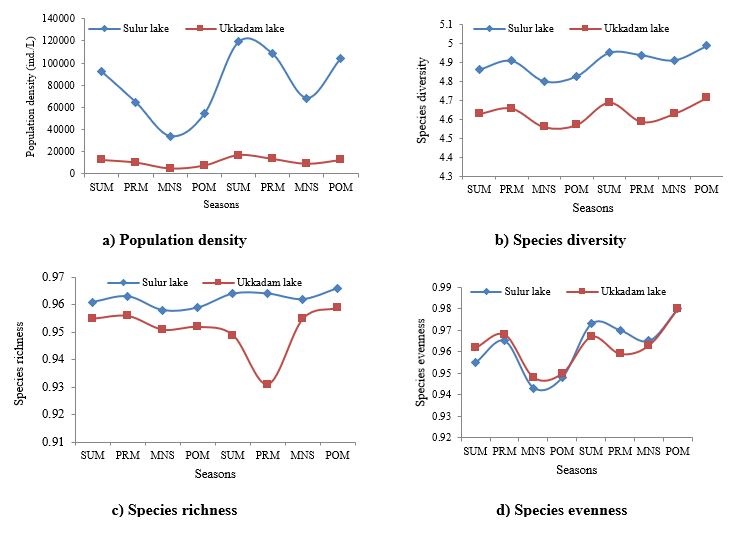
Population density (Quantitative analysis)
The population density of zooplankton recorded at Sulur and Ukkadam Lake was ranged between 51,895 and 1,89,435 ind./L (Figure 6a). At Sulur Lake zooplankton of population was recorded in the range between 51,895 and 1,07,505 ind./L. The minimum population was recorded during the monsoon season (September to November-2012), while the maximum during summer season (March to May-2013). In the Ukkadam lake, population density was reported in the range between 89,385 and 1,89,435 ind./L. The minimum population was recorded during the monsoon season (September to November-2012), while the maximum noticed during the summer season (March to May-2013).
Species diversity
Overall species diversity of zooplankton in Sulur and Ukkadam lake waters was recorded in the range between 4.56 and 4.99 (Figure 6b). In Sulur lake of species diversity was reported in the range between 4.80 and 4.99. The minimum diversity was recorded in monsoon season (September to November-2012) while maximum in post-monsoon season (December-2013 to February-2014). At Ukkadam lake species diversity was 4.56 - 4.71. The minimum diversity was obtained during monsoon season (September to November-2012), while maximum in post-monsoon season (December-2013 to February-2014).
Species richness
The overall species richness of zooplankton was ranged between 0.91 and 0.96 in Sulur and Ukkadam lake waters (Figure 6c). At the Sulur lake, species richness was 0.95 - 0.96. The minimum richness was reported in monsoon season (September to November-2012) and maximum in post-monsoon (December-2012 to February-2013). The species richness was noticed in the range between 0.93 and 0.96 in an Ukkadam lake. The minimum richness was recorded during the pre-monsoon season (June to August-2013), while the maximum in post-monsoon (December-2013 to February-2014).
Species evenness
The overall species evenness of zooplankton was 0.94 - 0.98 in Sulur and Ukkadam lake waters (Figure 6d). The species evenness was 0.94 - 0.98 with minimum recorded during the monsoon season (September to November-2012 and maximum during post-monsoon season (December-2013 to February-2014) at Sulur lake. The species evenness was noticed in the range between 0.95 and 0.98 at Ukkadam lake. The minimum evenness was recorded during the monsoon season (September to November-2012), while the maximum during post-monsoon season (December-2013 to February-2014).
|
Figure 6: Community structure of zooplankton in the Sulur and Ukkadam lake during March-2012 February-2014. Note: SUM - Summer; PRM - Pre-monsoon; MNS - Monsoon; POM - Post Monsoon. |
|
|
|
Figure 8: Percentage composition of zooplankton recorded in Ukkadam lake during March-2012 - February-2014. |
Discussion
The most of researcher have been reported on the rise in zooplankton species was richness in the lakes ecosystem [31] on size and with of lake depth [32]. The zooplankton is a major factor of aquatic ecosystem and changes in their abundance, species composition and seasonal variation have cascading effects at higher trophic levels [33]. Zooplankton community structure is influenced strongly by biotic-abiotic factor (water temperature, competition and predation) in freshwater ecosystem [34-37]. In the present study Sulur lake, results show the presences of 34 zooplankton species, comprising 11 Rotifera, 10 Cladocera, 7 Copepoda and 6 Ostracoda while in the Ukkadam lake it showed the presence of 28 species include 10 Rotifera, 8 Cladocera, 6 Copepoda and 4 Ostracoda. Rotifera group was greatest taxonomic richness. Similar results were obtained by several Indian researcher studies on [1,2,38-41] (Lakes and reservoirs, Dharmapuri and Krishnagiri District); [42] (Haledharmapuri lake, Dharmapuri, Tamil Nadu, India); [43] (Sulur Lake, Coimbatore District, Tamil Nadu, India).
Presently Cladocera and copepoda result were agreement with some earlier workers. [1,44-46] Some genera of copepoda and Cladocera are cosmopolitan in distribution, while others are restricted to some continents [47,48]. In the present study zooplankton percentage composition was found to be more by rotifera with 35% at Ukkadam and 34% at Sulur followed by Cladocera with 31% at Sulur and 30% Ukkadam copepoda with 27% (Ukkadam) and 25% (Sulur) and Ostracoda with 10% at Sulur lake and 8% of Ukkadam lake (Figure 8). The present result was analogous to earlier observation by Ramakrishna [49]; Manickam et al., [1,2,40,41]; Dede and Deshmukh [50]; Bhavan et al., [43]; Dhanasekaran et al., [42].
During the present study, overall population density of zooplankton was found maximum in summer season at both stations (Sulur and Ukkadam lake). In the summer season zooplankton population was found to be high which might be due to favorable environmental conditions and availability of food (phytoplankton) in lake environment. The distribution and population density of zooplankton in these water bodies depends upon the physicochemical parameters, the rich nutrient loading which supports the high phytoplankton productivity which can ultimately support the zooplankton population as agreed by Manickam et al., [1] from Thoppaiyar reservoir (Dharmapuri District, Tamil Nadu, India); Ramakrishna [49] from Yelahanka lake, (Bangalore, Karnataka, India); Gayathri et al., [51] from Doddavoderhalli lake, (Bangalore, Karnataka, India); Dede and Deshmukh [50] from Bhima river near Ramwadi Village (Solapur District, Maharashtra, India); Bhavan et al., [43] from Sulu lake (Coimbatore District, Tamil Nadu); Manickam et al., [40] from Barur Lake (Krishnagiri District, Tamil Nadu, India).
In the present study overall population density of zooplankton were found minimum during monsoon season in both stations which might be due to high turbidity, low light intensity, cloudy sky besides high rainfall as agreed by earlier works (Thirupathaiah et al., [52]; Manickam et al., [1,2,40,41]; Singh et al., [53]; Patel et al., [54]; Watkar and Bardate [55]; Ezhili et al., [56]; Dede and Deshmukh [50]; Bhavan et al., [43]; Dhanasekaran et al., [42]. In the present study zooplankton diversity was higher in Sulur lake than that of Ukkadam lake in summer season while lower in monsoon season as agreed by Kannan and Job [57]; Adesalu and Nwankwo [58]; Rajagopal et al., [59]. Although Balloch et al., [60]; Ismael and Dorgham [61] found that the diversity index (Shannon’s [29]) to be a suitable indicator for water quality assessment as the species diversity tends to be low in stressed and polluted ecosystem [62]. The maximum and minimum population of Cladocera in summer might be due to favorable temperature and availability of food in the form of bacteria, nanoplankton and suspended detritus while in monsoon the factors like water temperature, dissolved oxygen, turbidity and transparency play an important role in controlling the diversity and density of Cladocera [63]. The lake rich in organic matter support higher number of cyclopoids, thus suggesting their preponderance in higher trophic state of water.
During present study, zooplankton species richness in the Sulur lake was high in post-monsoon season during first year followed by Ukkadam lake in post-monsoon season during second year and lower levels in Sulur at monsoon season at first year as well as Ukkadam in pre-monsoon season of second years. The result was reported that the higher species richness is characterized by larger food chain [1,40,64]. In the present study zooplankton species evenness in the Sulur as well as Ukkadam lakes was high in post-monsoon season during second year and found low in Sulur during monsoon season in first year. Equitability (evenness) was relatively high during the rainy season indicating a reduction in the plankton diversity at this period [41]. Peet [65] has reported that species diversity implies both richness and evenness in the number of species and equitability for the distribution of individual among the species. The present study coincides with Odum [66] that the dominance value is inversely proportional to values of diversity, evenness and species richness. The results of seasonal variation in environmental parameters and plankton population suggest that the favorable period for primary production in Sulur and Ukkadam lake is October to November when nutrients accumulated from freshwater run-off due to monsoon rainfall in these months.
The temperature is an important factor in any environment affecting biological processes, the summer month showed high value temperature in Sulur and Ukkadam lake. Therefore, summer may be attributed to favorable temperature and availability of food in the form of bacteria, phytoplankton and suspended detritus while in monsoon the factors like water temperature, dissolved oxygen, turbidity and transparency play an important role in controlling the diversity and density of Cladocera. The zooplankton population shows sudden decrease during monsoon months indicates the fact that the prevailed physicochemical conditions were disfavoring for the growth of zooplankton because of the lentic water system. This effect may also be due to over predation of zooplankton by the higher trophic members such as planktivourous fishes, which regulating the zooplanktonic population in a water body [1,67]. The lowest zooplankton population recorded during monsoon may be related to low temperature [68].
Rotifera is one of the indicator organisms in the zooplankton groups. The species of B. calyciflorus considered to be a good indicator of eutrophication [1,38-40,42,43,59,69]. Among rotifera B. angularis, B. calyciflorus, F. longiseta and Lecane spp. indicate semi polluted waters [4,70], the dominance of Brachionus sp. and F. longiseta in the lake designate eutrophy and are usually recorded in mixotrophic waters [37,71,72]. In rotifera, species such as B. calyciflorus, B. falcatus and Filinia longiseta, some species of Cladocera (D. sarsi and C. cornuta), some species of copepoda (Heliodiaptomus viduus, Mesocyclops hyalinus and Thermocyclops hyalinus) and some species of Ostracoda (Cypris protubera and Hemicypris anomala) are the pollution tolerant species and indicate accumulation of organic matter and also reveal that the lake is being eutrophicated and polluted [1,2,39-41,43,73].
Habitually the monsoon months were associated with low zooplankton population due to dilution effect and decreased photosynthetic activity by primary producers. Similar results have been shown by Bais and Agrawal [74]. The population falls during the monsoon, due to a dilution effect of water turbulence. The population again rises to a higher level in winter, as a result of favorable environmental conditions. Normally, the monsoon is associated with lower population densities due to its dilution effect and decrease in photosynthetic activity by primary producers. Similar results have been shown by Edmondson [28], Baker [75], Bais and Agrawal [69], Salve and Hiware [76].
Furthermore, the relationship has been observed between a high number of the genus Brachionus and a high trophic level [77]. Thus, the quantitative and qualitative distribution and species composition of rotifers in Hulun lake indicated its highly eutrophic and polluted conditions. Moreover, rotifera prefer more alkaline waters [8,37]. The presence of five species of Brachionus indicated that the lake was approaching towards eutrophication and was semi-polluted. Different species of zooplankton population density showed their abundance according to the favorable condition, so they disappear in unfavorable conditions and reappeared on the return of favorable condition.
Conclusion
To concluded that the zooplankton population was maximum in summer season; the reason might be the favorable temperature and high levels of nutrients in lake and a high standing crop of primary producers leading to greater availability of food. Therefore, Ukkadam and Sulur lake was suitable for natural inland pisciculture (fin-fish and shell-fish) practices. In this given preliminary knowledge of information can be utilized to scientific community, policy maker for the effective conservation and managements measures to improve the water body in the lakes ecosystem.
Acknowledgments
Authors are thankful to the Head, Department of Zoology, Bharathiar University, Coimbatore, Tamil Nadu, India, for the necessary laboratory facilities provided. The first author (NM) gratefully acknowledged the SERB, Department of Science and Technology (DST), Govt. of India, New Delhi, for granting PI/National Post-Doctoral Fellowship (DST-SERB, File No.: PDF/2016/000738; Date - 05.06.2016) at Department of Marine Science, Bharathidasan University, Tiruchirappalli - 24, Tamil Nadu, India.
References
- Manickam N, Saravana Bhavan P, Santhanam P, Muralisankar T, Srinivasan V, et al. (2014) Seasonal Variations of Zooplankton Diversity in a Perennial Reservoir at Thoppaiyar, Dharmapuri District, South India. Austin Journal of Aquaculture and Marine Biology 1: 1-7.
- Manickam N, Santhanam P, Bhuvaneswari R, Saravana Bhavan P, Vijayadevan K, et al. (2017) Seasonal variations in zooplankton diversity and physico-chemical characteristics of the Nagavathi reservoir, Dharmapuri District, Tamil Nadu, India. Journal of Terrestrial and Marine Research 1: 28-35.
- Capriulo GM, Smith G, Troy R, Wikfors GH, Pellet J, et al. (2002) The planktonic food web structure of a temperate zone estuary, and its alteration due to eutrophication. Hydrobiologia 475: 263-333.
- Turner JT (2004) The importance of small planktonic copepods and their roles in pelagic marine food webs. Zool Stud 43: 255-266.
- Sotton B, Guillard J, Anneville O, Maréchal M, Savichtcheva O, et al. (2014) Trophic transfer of microcystins through the lake pelagic food web: evidence for the role of zooplankton as a vector in fish contamination. Sci Total Environ 1: 152-163.
- Zhao K, Wang L, Riseng C, Wehrlyd K, Pan Y, et al. (2017) Factors determining zooplankton assemblage difference among a man-made lake, connecting canals, and the water-origin river. Ecol Indi 84: 488-496.
- Bays JS, Crisman TL (2011) Zooplankton and trophic state relationships in Florida lakes. Can J Fish Aquat Sci 40: 1813-1819.
- Gannon JE, Stemberger RS (1978) Zooplankton (especially crustaceans and rotifers) as indicators of water quality. Trans Am Microsc Soc 97: 16-35.
- Pages F, Gonzalez HE, Ramon M, Sobarzo M, Gili JM (2001) Gelatinous zooplankton assemblages associated with water masses in the Humboldt Current System, and potential predatory impact by Bassia bassensis (Siphonophora: Calycophorae). Mar Ecol Prog Ser 210: 13-24.
- Jha P, Barat S (2003) Hydrobiological study of Lake Mirik in Darjeeling, Himalaya. J Environ Biol 24: 339-344.
- Abdulwahab S, Rabee AM (2015) Ecological factors affecting the distribution of the zooplankton community in the Tigris River at Baghdad region, Iraq. Egyptian Journal of Aquatic Research 41: 187-196.
- El-Bassat RA, Taylor WD (2007) The zooplankton community of Lake Abo Zaabal, a newly-formed mining lake in Cairo, Egypt. Afr J Aquat Sci 32: 185-192.
- Jeppesen E, Peder Jensen J, Sondergaard M, Lauridsen T, Landkildehus F (2000) Trophic structure, species richness and biodiversity in Danish lakes: changes along a phosphorus gradient. Freshw Biol 45: 201-218.
- Waters MN, Schelske CL, Brenner M (2015) Cyanobacterial dynamics in shallow Lake Apopka (Florida, USA) before and after the shift from a macrophyte dominated to a phytoplankton dominated state. Freshw Biol 60: 1571-1580.
- Binzer A, Guill C, Rall BC, Brose U (2016) Interactive effects of warming, eutrophication and size structure: impacts on biodiversity and food-web structure. Global Change Biol 22: 220-227.
- Zeng L, He F, Dai Z, Xu D, Liu B, et al. (2017) Effect of submerged macrophyte restoration on improving aquatic ecosystem in a subtropical, shallow lake. Ecological Engineering 106: 578-587.
- Fernández C, Estrada V, Parodi ER (2015) Factors triggering cyanobacteria dominance and succession during blooms in a hypereutrophic drinking water supply reservoir. Water Air Soil Pollut 226: 1-13.
- Mandal S, Goswami AR, Mukhopadhyay SK, Ray S (2015) Simulation model of phosphorus dynamics of an eutrophic impoundment East Calcutta wetlands, a Ramsar site in India. Ecol Model 306: 226-239.
- Xue Q, Steinman AD, Su X, Zhao Y, Xie L (2016) Temporal dynamics of microcystins in Limnodrilus hoffmeisteri, a dominant oligochaete of hypereutrophic Lake Taihu, China. Environ Pollut 213: 585-593.
- Ferreira JG, Bricker SB, Simas TC (2007) Application and sensitivity testing of a eutrophication assessment method on coastal systems in the United States and European Union. J Environ Manage. 82: 433-445.
- Van WJ, Ivens WP, Kroeze C, Löhr AJ (2015) Coastal eutrophication in Europe caused by production of energy crops. Sci Total Environ 511: 101-111.
- Conley DJ, Likens GE (2009) Ecology. Controlling eutrophication: nitrogen and phosphorus. Science 323: 1014-1015.
- Edmondson WT (1959) Freshwater Biology. 2ndedn, John Wiley and Sons Inc, New York, USA. Pg no: 1248.
- Sharma BK, Michael RG (1987) Review of taxonomic studies on freshwater Cladocera from India with remarks on biogeography. Hydrobiol 145: 29-33.
- Battish SK (1992) Freshwater Zooplankton of India. Oxford and IBH Publication Co. Pvt. Ltd, New Delhi, India. Pg no: 1-233.
- Murugan N, Murugavel P, Kodarkar MS (1998) Cladocera: The biology, classification, identification and ecology. Indian Association of Aquatic Biologists (IAAB) 5: 1-55.
- Altaff K (2004) A Manual of Zooplankton. Department of Zoology, The New College, Chennai. Tamil Nadu, India. Pg no: 19-145.
- Santhanam R, Velayutham P, Jegatheesan G (1989) A Manual of Freshwater Ecology. Daya Publishing House, Delhi, India. Pg no: 1-109.
- Shannon CE, Weaners W (1949) The mathematical theory of communications. University of Illinois, Urbana, USA. Pg no: 117.
- Gleason HA (1922) On the relation between species and area. Ecology 3: 156-162.
- Patalas K (1972) Crustacean plankton and the eutrophication of Lawrence Great Lakes. J Fish Res Bd Can 29: 1451-1462.
- Keller W, Conlon M (1994) Crustacean zooplankton communities and Lake morphometry in Precambrian shield Lakes. Cana J Fisher Aqua Sci 51: 2424-2434.
- Khalifa N, El-Damhogy KA, Fishar MR, Nasef AM, Hegab MH (2015) Vertical distribution of zooplankton in Lake Nasser. Egyptian Journal of Aquatic Research 41: 177-185.
- Dumont HJ (1994) On the diversity of Cladocera in the tropics. Hydrobiologia 272: 27-38.
- Karl C, Nele N, Erik M, Luc DM (2001) Zooplankton community structure and environmental conditions in a set of interconnected ponds. Hydrobiologia 442: 339-350.
- Meerhoff M, Clemente JM, Teixeira F, Mello F, Iglesias C, et al. (2007) Can warm climate-related structure of littoral predator assemblages weaken the clear water state in shallow lakes. Global Change Biology 13: 1888-1897.
- Ana XP, Dub ZH, Zhang JH, Li YP, Qi JW (2012) Structure of the zooplankton community in Hulun Lake, China. Procedia Environmental Sciences 13: 1099-1109.
- Manickam N, Saravana Bhavan, P, Santhanam P, Chitrarasu, P, Jawahar Ali A (2012) Zooplankton diversity in a perennial freshwater lake. Diversity and Physiological Processes. Department of zoology, Goa University, Goa, India. Pg no: 25-37.
- Manickam N, Saravana Bhavan P, Vijayan P, Sumathi G (2012) Phytoplankton species diversity in the Parambikulam-Aliyar irrigational canals Tamil Nadu, India. International Journal of Pharma and Bio Sciences 3: 289-300.
- Manickam N, Saravana Bhavan P, Santhanam P, Muralisankar T, Srinivasan V, et al. (2015) Biodiversity of freshwater zooplankton and physico-chemical parameters of Barur Lake, Krishnagiri District, Tamil Nadu, India. Malaya Journal of Bioscience 2: 1-12.
- Manickam N, Saravana Bhavan P, Santhanam P (2017) Evaluation of nutritional profiles of wild mixed zooplankton in Sulur and Ukkadam Lakes of Coimbatore, South India. Turkish Journal of Fisheries and Aquatic Sciences 17: 509-517.
- Dhanasekaran M, Saravana Bhavan P, Manickam N, Kalpana R (2017) Physico-chemical characteristics and zooplankton diversity in a perennial lake at Dharmapuri (Tamil Nadu, India). Journal of Entomology and Zoology Studies 5: 285-292.
- Bhavan PS, Selvi A, Manickam N, Srinivasan V, Santhanam P, et al. (2015) Diversity of Zooplankton in a pe-rennial Lake at Sulur, Coimbatore, India. Int J Ext Res 5: 31-44.
- Raghunathan MB (1983) Studies on some planktonic Cladocerans (Crustacea) of Tamil Nadu, Ph.D. Thesis, University of Madras, India.
- Sharma BK (1991) Cladocera: Animal Resources of India. Pg no: 205-223.
- Sivakumar K, Altaff K (2004) Ecological Indices of Freshwater Copepods and Cladocerans from Dharmapuri District, Tamil Nadu, India. Zoos’ Print Journal 19: 1466-1468.
- Brooks JL (1959) Cladocera. In: Freshwater Biology, 2nd Ed. Edmondson WT (ed.). John Wiley & Sons. New York, USA, Pg no: 587-656.
- Williamson CE (1991) Ecology and classification of North American freshwater invertebrates. In: Thorp JH, Covich AP (eds.). Academic Press Inc. San Diego, USA. Pg no: 787-822.
- Ramakrishna S (2014) Zooplankton seasonal abundance in relation to physicochemical feature in Yelahanka Lake, Bangalore, India. Global J Res Anal 3: 218-219.
- Dede AN, Deshmukh AL (2015) Study on Zooplankton Composition and Seasonal Variation in Bhima River Near Ramwadi Village, Solapur District (Maharashtra), India. Int J Curr Microbiol App Sci 4: 297-306.
- Gayathri S, Latha N, Mohan MR (2014) Studies on population dynamics and seasonal abundance of zooplankton community in Doddavoderahalli Lake, Bangalore, India. Int J Emerg Trends Eng Dev 4: 50-55.
- Thirupathaiah M, Sammatha CH, Sammaiah CH (2011) Diversity of zooplankton in freshwater Lake of Kamalapur, Karimnagar District, Andhra Pradesh, India. Ecoscan 5: 85-87.
- Singh RK, Pandey MK, Kumari R, Ranjan P (2012) Study on the diversity and seasonal variation of zooplankton in Mahendra Nath pond, Siwan, Bihar. Int J Pharm Biol Arch 3: 867-870.
- Patel V, Shukla SN, Patel VK (2013) Studies on the diversity of zooplankton and their seasonal variation in Govindgarh Lake at Rewa, Madhya Pradesh, India. Ind J Appl Res 3: 544-546.
- Watkar AM, Barbate MP (2013) Studies on Zooplankton Diversity of River Kolar Saoner, Dist- Nagpur, Maharashtra. J Life Sci Technol 1: 26-28.
- Ezhili, N, Manikandan R, Ilangovan R (2013) Diversity and seasonal variation of zooplankton in Ukkadam Lake, Coimbatore, Tamil Nadu, India. Int J Cur Res 8: 2091-2094.
- Kannan V, Job SV (1980) Diurnal, seasonal vertical study of primary production in Sathiar Reservoir. Hydrobiologia 70: 171-178.
- Adesalu, TA, Nwankwo DI (2008) Effect of water quality Indices on phytoplankton of a sluggish tidal creek in Lagos, Nigeria. Pak J Biol Sci 11: 836-844.
- Rajagopal T, Thangamani A, Sevarkodiyone SP, Sekar M, Archunanl G (2010) Zooplankton diversity and physico-chemical conditions in three perennial ponds of Virudhunagar district, Tamilnadu. J Envir Biol 31: 265-272.
- Balloch D, Davies CE, Jones FH (1976) Biological Assessment of Water Quality in three British Rivers: The North Esk (Scotland), the level (England) and the Taff (Wales). Water Poll Cont 75: 92-114.
- Ismael AA, Dorgham MM (2003) Ecological indices as a tool for assessing pollution in El- Dekhaila Harbor (Alexandria, Egypt). Oceanologia 45: 121-131.
- Bass D, Harrel RC (1981) Water quality of South East Texas Stream. Hydrobiol 76: 69-79.
- Edmondson WT (1965) Reproductive Rate of Planktonic Rotifers as Related to Food and Temperature. Ecol Monogra Orginat 35: 61-111.
- Dumont HJ (1999) The Species Richness of Reservoir Plankton and The Effect of Reservoir on Plankton Dispersal (with Particular Emphasis on Rotifers and Cladocerans). In: Tundisi JG, Straskraba M (eds.). Theoretical reservoir ecology and its applications. Internat Inst Ecol, Sao Carlos. Pg no: 477-492.
- Peet RK (1974) The measurement of species diversity. Ann Rev Ecol Syst 5: 285-307.
- Odum EP (1983) Basic Ecology. Holt-Saunders International Editions, Saunders College Publishing, Japan.
- Poongodi R, Bhavan PS, Vijayan P, Kannan S, Karpagam S (2009) Population of zooplankton in relation to physico-chemical parameters of a seasonal pond. Res Envir Lif Sci 2: 105-110.
- Marshall SM, Orr AP (1972) The charophytes of the Dal Lake, Kashmir. Abstract Proceeding. 21st Land Congress 295.
- Sampaio E, Rocha T, Tundisi M, Tundisi J (2002) Composition and abundance of zooplankton in the limnetic zone of seven reservoirs of the Paranapanema River. Brazil J Biol 62: 525-545.
- Vasquez E, Rey J (1992) Composition, abundance and biomass of zooplankton in Orinoco floodplain lakes, Venezuela. Annals Limnology 28: 3-11.
- Saksena DN (1987) Rotifers as indicators of water quality. Acta Hydrochimica et Hydrobiologia 5: 481-485.
- Patrick R (1950) Biological measure of stream conditions. Sewage Industrial Wastes 22: 926-938.
- Goel PK, Charan VR (1991) Studies on the liminology of polluted fresh water tank. In: Gopan B, Asthana V (eds.). Aquatic Sciences in India. Ind Ass Lim Oce Pg no: 51-64.
- Bais VS, Agrawal WL (1993) Seasonal Variation of Nutrient content in Hydrilla verticillata. J Freshwat Biol 3: 259-265.
- Baker RL (1979) Specific status of Keratella Cochlearis and K. Earlinare, Ahlstrom (Rotifera: Brachionidae): Morphological and ecological consideration. Canad J Zool 57: 1719-1722.
- Salve B, Hiware C (2010) Zooplankton diversity of Wan reservoir, Nagapur (Madhya Pradesh), India. Tree Res Sci Tech 2: 39-48.
- Frutos SM, Neiff PD, ASG, Neiff JJ (2009) Zooplankton abundance and species diversity in two lakes with different trophic states (Corrientes, Argentina). Acta Limnol Bras 21: 367-375.
 LOGIN
LOGIN REGISTER
REGISTER.png)